NEWS
Credibility and confidence
November 8, 2024
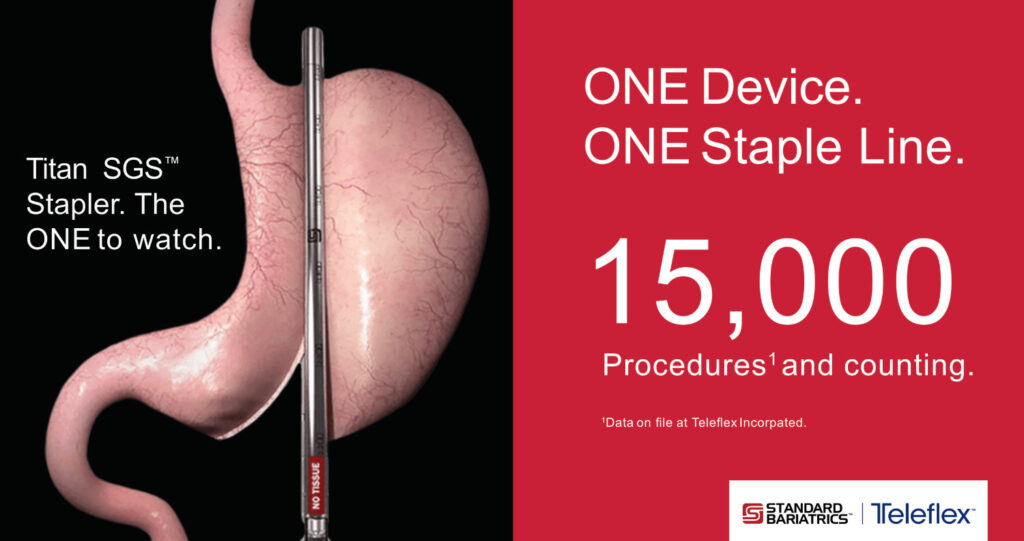
Surgeons have completed more than 15,000 sleeve gastrectomy procedures using the Titan SGS™ Stapler.1
1.Data on file at Teleflex Incorporated.

New study reports perioperative outcomes of the Titan SGS™ Stapler
November 1, 2024
Here’s exciting news for patients and surgeons: a study published in Obesity Surgery in August 2024 links our Titan SGS™ Stapler to potentially enhanced perioperative outcomes compared to multi-fire surgical staplers.

Standard Bariatrics® Titan SGS Stapling Technology for Sleeve Gastrectomy Receives FDA Clearance
Standard Bariatrics® announced today that its novel Titan SGS™ stapling technology, designed specifically for bariatric sleeve surgery, has been cleared by the US Food and Drug Administration (FDA). The Titan SGS™ first-of-its-kind design offers surgeons performing sleeve gastrectomy procedures the industry’s longest continuous staple cutline of 23 centimeters.

Standard Bariatrics to attend ObesityWeek 2019
Come visit us in Booth #1014 at ObesityWeek 2019 in Las Vegas, NV from November 3rd through 7th.

2019 Innovation & Technology Awards winner: Standard Bariatrics
Standard Bariatrics is developing products to improve the consistency and clinical outcomes of sleeve gastrectomy, the surgical procedure most commonly used in the United States to treat morbid obesity.

Standard Bariatrics Receives FDA Indication for #1 Bariatric Surgical Procedure
Standard Bariatrics, an innovator of surgical devices, has received a sleeve gastrectomy indication from the Food and Drug Administration (FDA) for the STANDARD CLAMP®.

Standard Bariatrics to attend ObesityWeek 2018
Come visit us in Booth #1103 at ObesityWeek 2018 in Nashville, TN from November 11 through 15th.
Learn more about ObesityWeek 2018.

Standard Bariatrics receives 2018 Innovation & Technology Award
Standard Bariatrics received the 2018 Innovation & Technology Award in the category of Outstanding Bioscience from the Business Courier‘s eighth-annual Innovation & Technology Awards, recognizing companies that are fueling growth through innovative processes and products.
Read the full article on Cincinnati Business Courier
